I often criticize the U.S. Food and Drug Administration (FDA) for being stodgy, slow to react and overly risk averse. However, the agency has an important purpose. The forerunner of the FDA dates back to the 1906 Pure Food and Drugs Act. At the time unscrupulous food manufacturers would use red dye to make spoiled beef appear fresh and used formaldehyde as a preservative to make foods last longer. Only a few years prior, Heroin was considered a wonder drug, as a treatment for morphine addiction and as a cough suppressant. Morphine was also once used as an elixir for teething pain in babies.
In the 1800s, Mrs Winslow’s Soothing Syrup was allegedly responsible for the death of babies in America. Marketed a ‘miracle cure’ to ease the pain of teething tots, parents didn’t realise the syrup contained morphine and it’s thought many infants died of an overdose. So-called ‘medicines’, featuring morphine and opium, were often packaged in pretty bottles and given a quaint, home-remedy name.
In 1960 FDA medical reviewer, Frances Oldham Kelsey, famously refused to approve Thalidomide, claiming safety data was lacking. The drug was later tied to severe birth defects and Dr. Kelsey became a rock star at the FDA. Kelsey retired from the FDA at age 90 after 45 years of service. She averted a public health calamity but the FDA took her example to heart, perhaps a little too much.
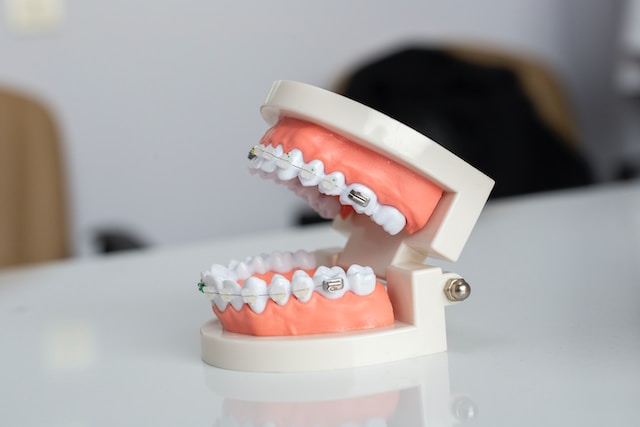
Besides foods and drugs, the FDA also regulates medical devices. An anterior growth guidance appliance (AGGA) dental device has been in the news lately. The Justice Department recently announced a criminal investigation into its developer. The device was designed to expand the jaw, lengthening it over time. It is being sold to fix various problems from cosmetic applications, like a weak chin, to alleviating jaw pain, to a cure for temporomandibular joint (TMJ) syndrome. Despite being distributed through dentists it was never submitted to the FDA for review. A Kaiser Health News investigation found its users experienced numerous problems, including pushing teeth away from bone rather than forcing their jawbone to expand. According to Kaiser Health News:
More than 10,000 dental patients have been fitted with an Anterior Growth Guidance Appliance, or “AGGA,” according to court records. But the unproven and unregulated dental device, often costing patients about $7,000, has not been evaluated by the FDA, according to a months-long joint investigation by KHN and CBS News. The FDA relies on device companies to submit new products for evaluation, and because the AGGA was never submitted, it has been sold to patients without that government review.
The device is still on the market despite the problems experienced by patients. Patients are actually losing teeth after teeth were pushed away from bone. Developed by a dentist nearly 30 years ago, the science behind it sounds implausible.
It is well established that teeth will move within the jaw when gently forced through small increases in pressure. The science of moving teeth within the jaw is called orthodontics. I had braces as a teen. Over the course of a year or two my braces slowly moved wayward teeth into alignment. The AGGA inventor apparently had the idea that if you anchor the device to the back molars and push against the front teeth using two springs, it would encourage the jawbone to stretch, growing forward. This theory actually works when expanding leg bones to make people taller. However, the leg bones first have to be broken. Bone growth occurs along the break as the break is slowly pulled apart and the bone tries to heal the break. In the case of the dental device, what apparently happened in some cases was rather than forcing the jaw to expand the device moved teeth forward until they were no longer anchored with enough bone to hold them in place.
I found this account interesting because of an earlier case of a dental TMJ product that harmed numerous patients. More than 30 years ago Vitek, Inc. from Houston, Texas manufactured dental implants to alleviate TMJ. These devices were made of a porous polytetrafluoroethylene-based material (i.e. Teflon). Teflon is the nonstick plastic coating on old, nonstick frying pans that scrapes off over time. It is not a resilient coating when using metal cooking utensils and apparently not a material durable enough to chew with either. Under the constant pressure of the jaw Teflon-based implants broke down. The result was pain and disfigurement, but often symptoms didn’t manifest until it was too late to reverse. The following is from a 30-year old article in the Washington Post.
…the jaw implants can break down under pressure, damaging adjacent bones and in some cases perforating the skull.
“These implants are deteriorating, often without any clinical symptoms such as pain or disfigurement. When the pain and restricted jaw movement do appear, the unseen damage is well advanced,” said Gerald Kuester, a researcher for the group.
Early last year FDA ordered the manufacturer, Vitek Inc. of Houston, to notify all oral surgeons to whom implants were sold. But in June, Vitek declared bankruptcy before contacting all the surgeons.
There were 26,000 Vitek TMJ implants sold, of which about 10,000 were installed before the FDA managed to seize the remaining inventory just as the inventor was trying to take it and flee the country. According to the LA Times:
[The] …quarter-sized, doughnut-shaped disc made of a material called Proplast Teflon, a porous, sponge-like plastic fused with carbon or aluminum oxide.
But it turned out the man-made implants wore out too, crumbling into microscopic sand-like grains of Teflon, which the body can’t metabolize. That created what scientists call a “giant cell” reaction, where the body tries to engulf the foreign matter, triggering inflammation.
The reaction can continue after implants have been removed and replaced, even with a patient’s own tissue.
In the late 1980s extensive clinical testing of medical devices was not required. The Vitek implants had been cleared to market by the FDA, using a process called 510(k), where new products are shown to work similar to existing medical device products on the market. Even if the products worked similarly to other products on the market, the Proplast Teflon did not hold up and was ultimately forced off the market by the FDA. The company regrouped in Switzerland but later closed there too.
Coca-Cola.
Yes, and lot of other products that contained alcohol and opium to created, satisfied, repeat customers. The list of maladies these elixirs would cure guaranteed you could use them for anything.